 
This approach, entailing the prospective screening and selection of integration sites, based on combined bioinformatics and functional analyses, provides a robust and dependable strategy for the genetic engineering of human iPS cells. In a recent proof-of-principle study, using a model for genetic correction of β-thalassemia major, we demonstrated that erythroid progeny of patient-specific iPS cell clones harboring a lentivirally encoded β-globin transgene in a safe harbor site express therapeutic levels of β-globin without perturbing neighboring genes. To test them, we developed a strategy to select iPS cell clones harboring a single copy of a randomly integrating vector at sites that meet our safe harbor criteria. We proposed a definition of safe harbor sites, based on their topology in the genome with relation to coding genes and other genomic landmarks, using five criteria: (i) distance of at least 50 kb from the 5’ end of any gene, (ii) distance of at least 300 kb from any cancer-related gene, (iii) distance of at least 300 kb from any microRNA, (iv) location outside a transcription unit, and (v) location outside ultraconserved regions of the human genome ( 1). iPS cells, unlike HSC, can be extensively cultured in vitro, enabling the selection and study of unique sites of transgene integration for the first time in a relevant setting. This risk would be minimized if therapeutic transgenes could be inserted in selected sites of the genome that permit appropriate function without disruption or dysregulation of endogenous genes - referred to as “genomic safe harbors.” The advent of induced pluripotent stem (iPS) cell technology offered unprecedented opportunities for the genetic engineering of human cells. 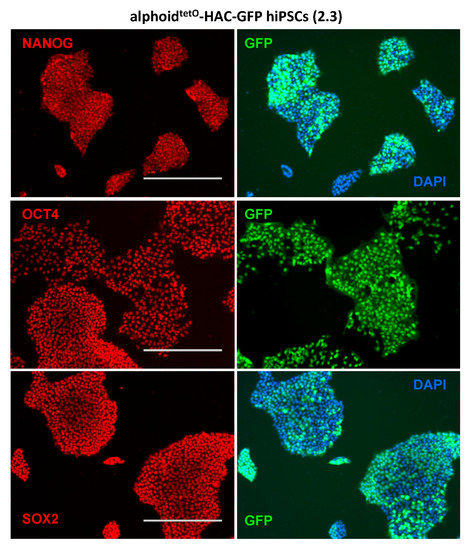
Molecular mechanisms underlying pluripotencyĥ.Current hematopoietic stem cell (HSC) gene therapy relies on randomly integrated retroviral vectors and is hampered by the risk of insertional oncogenesis often leading to leukemia. Understand cellular features that sustain pluripotency ĭevelop tools to evaluate stem cell potential Ĭompare different pluripotent stem cells and strategies for obtaining them Įvaluate which diseases and conditions can be modeled using iPS cellsĮvaluate the use of different types of pluripotent stem cells in experiments or in the clinic.ġ.Ğstablishment of embryonic stem cells (ESCs)Ģ.Ĝharacterization of pluripotent stem cells (PSCs)ģ. Identify defining pluripotent stem cell features ĭesign experiments to test if cells are pluripotent Additionally, this module contains an extra set of topics for advanced students interested in pluripotent stem cell biology.Īt the conclusion of this module, students should be able to: The module ends with an overview of the possible clinical application of pluripotent stem cells as a window into their medical and research potential. Understanding pluripotency will unlock reprogramming and the generation of induced pluripotent stem cells. There is a special emphasis on defining pluripotency by molecular means and differentiation potential. Students will learn what an embryonic stem cell is, how it is derived, and what makes them pluripotent. Module 3 dives into different types of pluripotent stem cells. Module 3 | Pluripotency and Reprogramming in vitro Introduction and Learning Objectives: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
